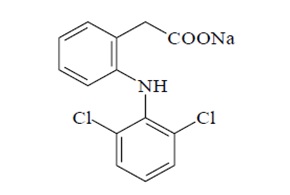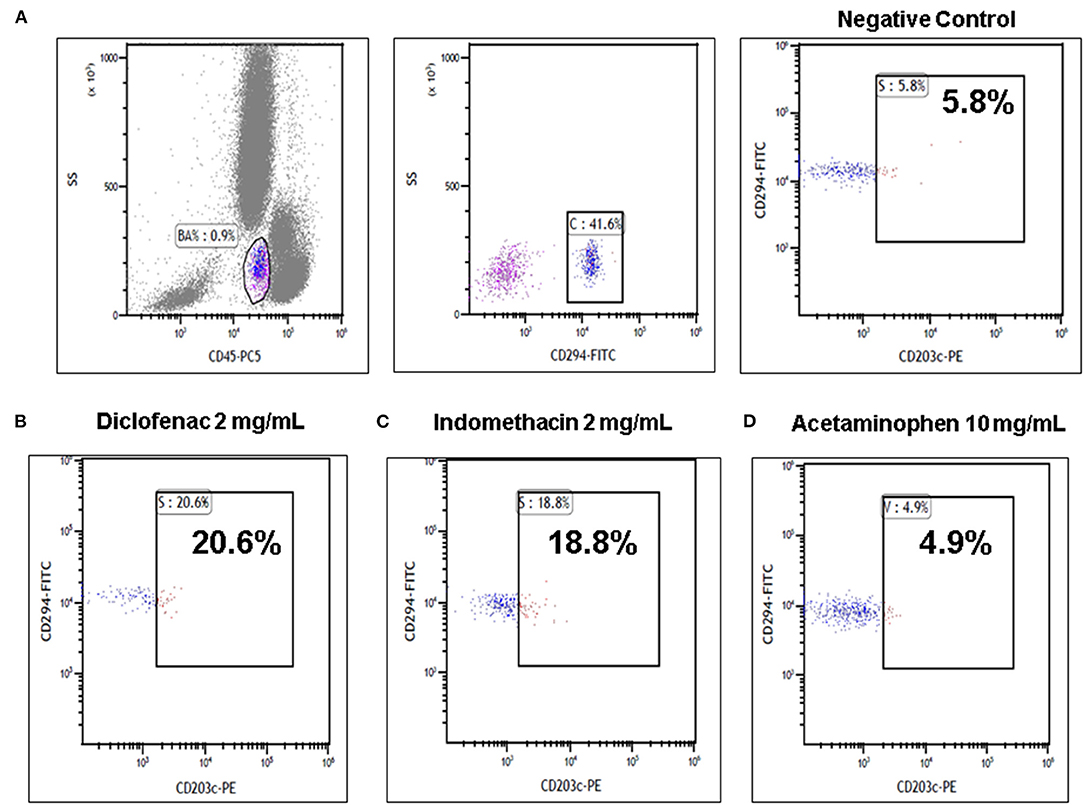
Frontiers | A Case Report of a 3-Year-Old Child With Anaphylactic Shock After a Diclofenac Suppository Confirmed by Serial Tryptase and a Basophil Activation Test

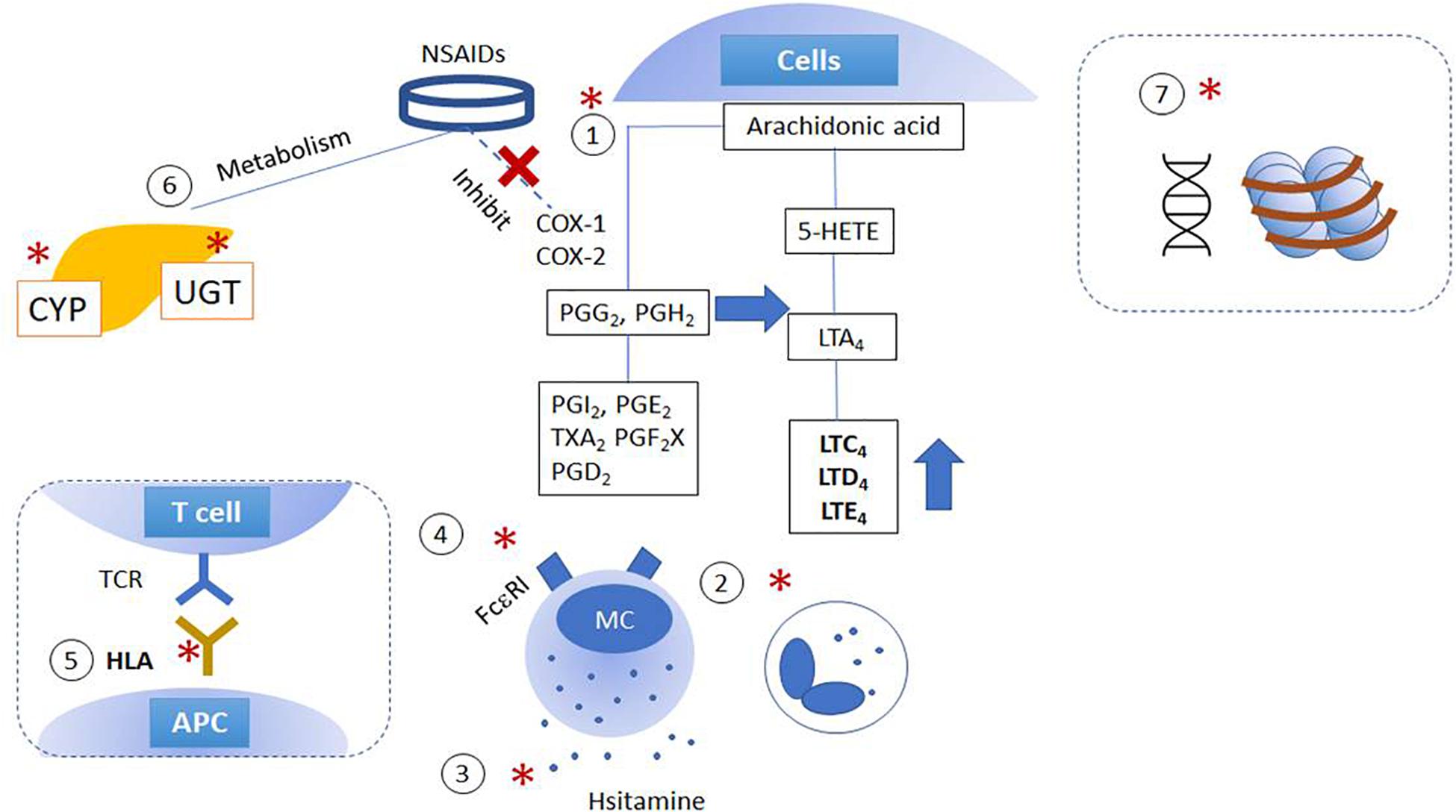
Progress in understanding hypersensitivity reactions to nonsteroidal anti‐inflammatory drugs - Doña - 2020 - Allergy - Wiley Online Library

Toxics | Free Full-Text | Diclofenac Concentrations in Post-Mortem Specimens—Distribution, Case Reports, and Validated Method (UHPLC-QqQ-MS/MS) for Its Determination

Frontiers | Non-steroidal anti-inflammatory drug-induced anaphylaxis infrequent in 388 patients with mastocytosis: A two-center retrospective cohort study
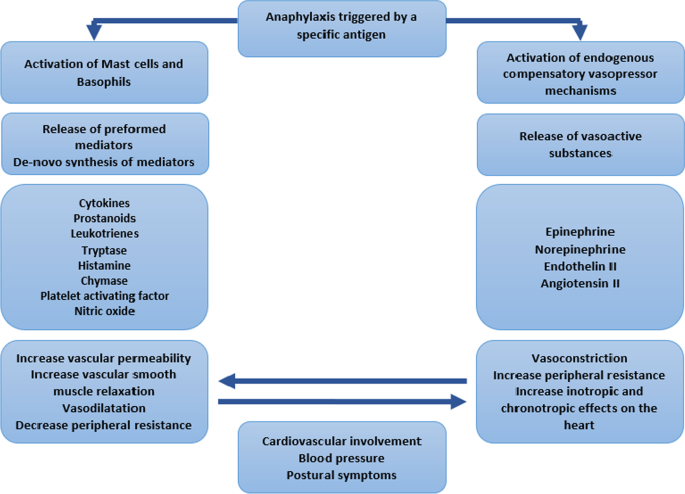
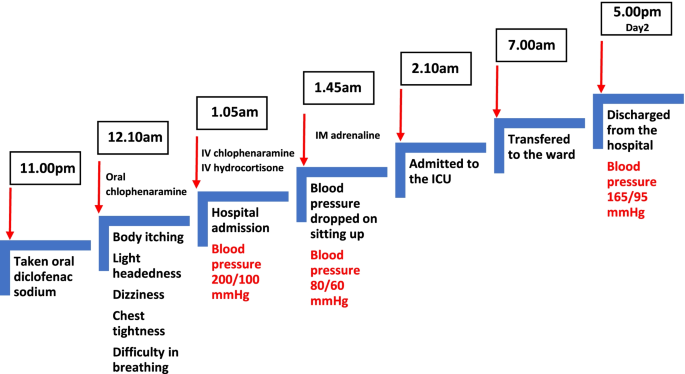
An unusual presentation of anaphylaxis with severe hypertension: a case report | Journal of Medical Case Reports | Full Text
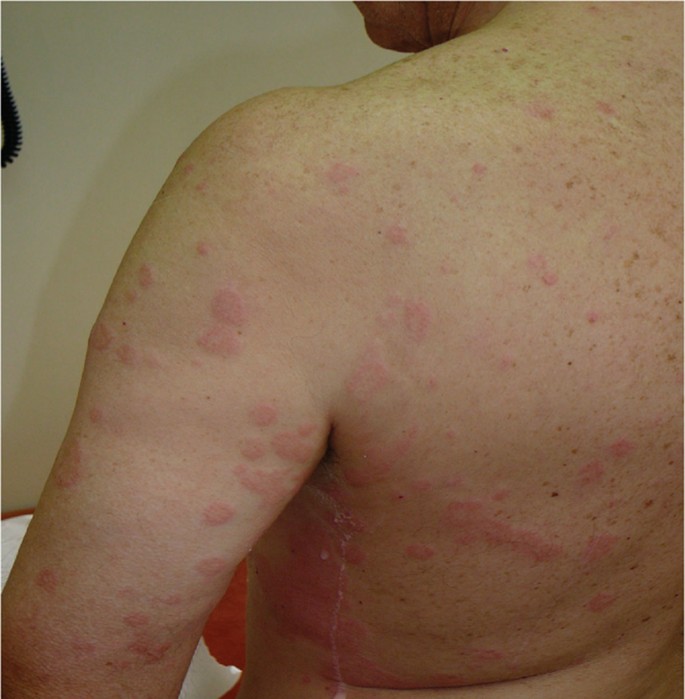
Clinical Management of Nonsteroidal Anti-inflammatory Drug Hypersensitivity | World Allergy Organization Journal | Full Text

Hypersensitivity reactions to non-steroidal anti-inflammatory drugs: results of an Austrian cohort study | Allergo Journal International

Emerging Causes of Drug-Induced Anaphylaxis: A Review of Anaphylaxis-Associated Reports in the FDA Adverse Event Reporting System (FAERS) - ScienceDirect

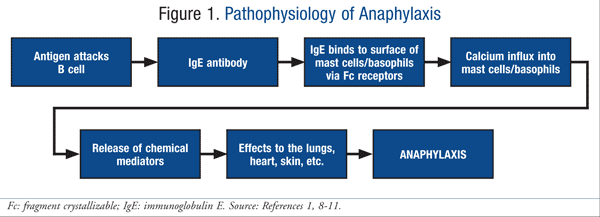
Characterization of anaphylaxis reveals different metabolic changes depending on severity and triggers - Perales‐Chorda - 2021 - Clinical & Experimental Allergy - Wiley Online Library

PDF) A Case Report of a 3-Year-Old Child With Anaphylactic Shock After a Diclofenac Suppository Confirmed by Serial Tryptase and a Basophil Activation Test